Part 2: Boron Based Acids
Introduction
In the first year (at Imperial anyway) the Lewis acidity of various compounds is discussed. Lewis acidity being the ability of a substance to act as an electron pair acceptor. Boron compounds of the type BX3 (we are taught) are electron deficient, the B atom contributes 3e to its valence shell and then each "X" contributes another e, giving a total of 6e, when really 8e are required to reach a full octet, and hence boron compounds readily accept additional electrons.
This being the case, where X=halide we would expect the acidity of BX3 compounds to decrease from BF3 > BCl3 > BBr3 since, this is the order of their electronegativity, F will withdraw more electron density from the B center making it even more Lewis acidic.
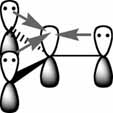 However, the Lewis acidity of these compounds progresses as BBr3 > BCl3 > BF3. The explanation for this reversal of the expected trend is placed on the p-AOs of the halide, these orbitals can form a ą-like interaction with the boron atom, where the lone pairs on the halide back-donate electron density into the empty pAO on the boron.
However, the Lewis acidity of these compounds progresses as BBr3 > BCl3 > BF3. The explanation for this reversal of the expected trend is placed on the p-AOs of the halide, these orbitals can form a ą-like interaction with the boron atom, where the lone pairs on the halide back-donate electron density into the empty pAO on the boron.

However, even this hypothesis has been downgraded and molecular orbital arguments have been used to discuss the relative acidity of boron based acids. BCl3 is inherently a stronger acid than BF3, it forms a stronger B-X bond and a more stable adduct with NH3. This is in part due to the LUMO of BCl3 lying closer in energy to the HOMO of NH3 resulting in a stronger interaction between these orbitals.
In this part of the lab your job is to investigate the boron based acids BF3 and BCl3, and their interaction with the lewis base NH3.
Key tasks are to:
- Optimize structures of BF3, BCl3 and NH3 individually
- Perform a frequency analysis on all structures to confirm the minimum and obtain the vibrational frequencies
- Critically analyse the bonding of BF3, BCl3 relative to BH3 , you will need to use the following input files for the NBO analysis: BF3 input file and BCl3 input file
- Optimize structures of the adduct F3B:NH3, and then carry out the frequency analysis
- Critically analyse the bonding in the adduct.
Here you will want to refer to the tutorial on BH3 and to examine the extent to which BF3 becomes non-planar due to partial occupation of the perpendicular pAO and mixing effects becoming active. - If you have time compare the adduct F3B:NH3 with those for H3B:NH3 and Cl3B:NH3
- Alternatively you could explore the effect of other substituents on either the N or B, for example in BR3 replace R with pyridine, OH, NH2 or CH3, or in NR3 replace R with F ... what effect would you expect these substituents have on the extent of lone pair donation from the N to the B? Is your hypothesis consistent with your calculated results?
