Ionic Liquids Research
Areas of intrest and research including current projects
Research spans a number of areas which are linked by an interest in inorganic bonding, catalysis, reactivity measures and combustion processes. This involves determining a mechanistic profile for a chemical reaction and examining key structures in the process.We develop an understanding of the underlying electronic structure which drives the reactivity and then predict and explore how chemically modification of the reactants or catalyst will facilitate the primary catalytic reaction, and also retard side reactions.
Very often this will require an in depth understanding of complex chemical bonding, moving beyond simple modes of representation into a detailed understanding of the molecular orbitals, charges and electronic distribution.
The following topics include current areas of active research and new project areas. Click on a link, or scroll down to see each topic.
Solvent assisted frustriated lewis pair clevage of H2
Frustrated Lewis pairs are acids and bases which want to form a dative bond (acid-base adduct) but which are hindered from doing so by particularly bulky substitents. This results in a very long and chemically active dative bond, which can be exploited for main group catalytic activity. The most common acids are ER3 where E=B or Al, and the most common bases are ER3 where E=N, P. We are studying are the novel silylium ions R3Si+ acids combined phosphane bases. These compounds do succeed in forming a tranditional adduct, however on solvent coordination the adduct bond is lengthened forming a solvent assisted FLP which is capable of cleaving H2.
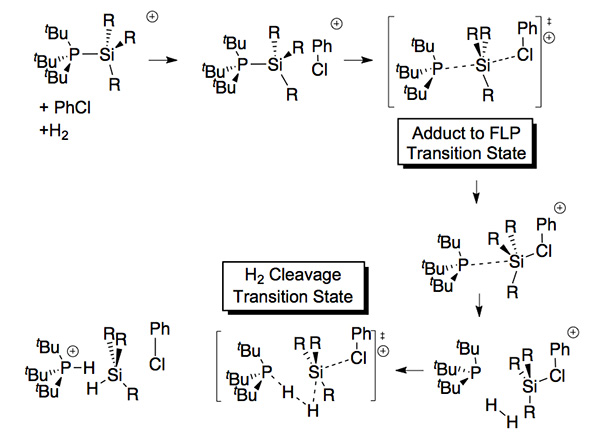
FLP assisted mechanim for H2 clevage
Group II catalytic hydroamination
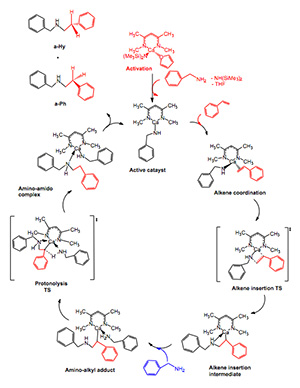
Hydroanimation is the catalytic addition of an N-H bond to an unsaturated C-C multiple bond. Such carbon-nitrogen bond forming processes are one of the key reactions in organic chemistry. For example, this reaction is used in the production of N-containing polycyclic frameworks for natural product synthesis. We are involved with research to produce alkaloids, these are used pharmaceutically to control bleeding, alleviate pain, reduce fever, and as anesthetics and antibiotics.
Group 2 metals are not your normal candidates for catalysis, they are often percieved as inert counter ions. However, group II metals can catalise the hydroanimation of an olefin to produce aminoalkanes. We have completed a study of the catalytic cycle for a simple model of ammonia with ethene, and are now characterising the cycle for activated alkenes (reaction shown on the left)
Our experimental collaborator Dr Hill has discovered that Ca catalyses hydrophosphination as well hydroamination, however the character of the mechanism shows significant differences, for example for some substrates polymerization and not hydrophosphination occurs and we are investigating why!
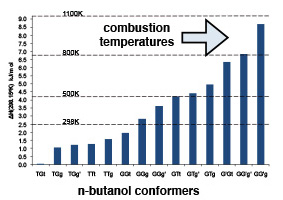
H abstraction from n-butanol by the methyl radical
n-Butanol (biobutanol) is an attractive biofuel because it has a higher energy content than ethanol, can be easily blended with conventional gasoline in higher concentrations without engine modification, produces less smog and is less suseptible to separation in the presence of water.
H-abstraction reactions are important pathways in the high-temperature decomposition of fuels because they provide the necessary pool of radicals to sustain the combustion process. Reaction flow analysis has revealed that H-abstraction by the methyl radical is of increased importance during pyrolysis because of the very low concentration of other radical species. We have studied the H-abstraction reactions of the methyl radical determining barrier heights, product stabilities, kinetics and reaction rates.
Most studies of pyrolysis focus on the lowest energy conformer, however at the elevated temperatures at which the fuel burns higher energy conformers are present in siginficant proportions. We have studied the combustion reaction profiles for these species in comparison to the lowest energy confomer.
doi Hydrogen Abstraction from n-Butanol by the Methyl Radical: High Level Ab Initio Abstraction Pathways and the Importance of Low Energy Rotational Conformers, Patricia Hunt, Alex Taylor, Yannis Hardalupas and Dimitrios Katsikadakos, PCCP, 2012, 14, 9615-9629