The Effective Molecular Method (EMO) Method
When a molecule is introduced into an aqueous solvent, the water molecules arrange themselves about the solute in a "solvent shell". This solvent shell is not static and the solvent molecules move around, and even enter and exit the solvent shell over time.
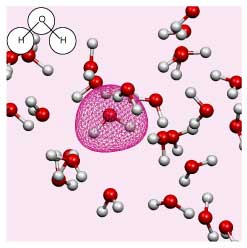
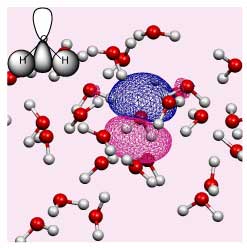
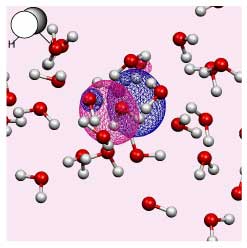
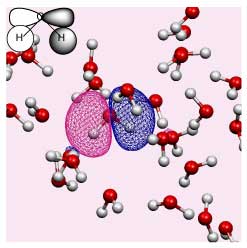
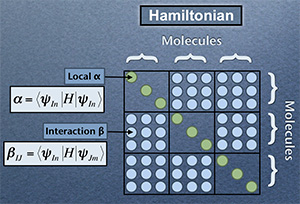
The changing environment impacts on the electronic structure of the solute and solvent. However, the Kohn-Sham orbitals obtained for a solute in liquid water are highly delocalised over all the molecules in solution (see the figure on the right).
Until now it has been impossible to isolate information pertinent to a specific solute. We have developed a new method for extracting effective solute MOs, as well as for analyzing local individual solute solvent interactions via a beta parameter.
This takes us a large way forward in the study of solvation effects, and gives us ability to distinguish and investigate the reactivity of solutes in solution. We are now able to examine the molecular orbitals (MOs) of molecules in solution, and to examine how they change over time.

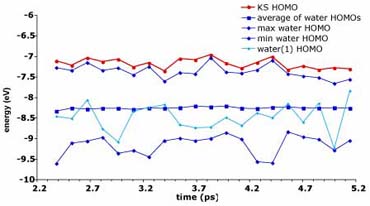
In this graph the light blue line represents the change in energy of the HOMO (highest occupied orbital) of one water molecule over time. This water molecule represents a "solute", and the remaining water molecules the "solvent". The "solvent" HOMOs form a band of energies which lie between the two sets of blue diamond marked lines.